What is membrane filtration?
During the last decades membrane filtration has taken an increasingly important role in the water treatment's framework, it's considered the technology of the future.
This success is due to the fact that it grantees an efficient filtration, but at the same time it's eco friendly and with a minimal use of chemical products. Membrane filtration is based on a simple physical process: the membranes are constituted by thin layers of semi selective material that behaves as a “sieve" through which the water is pushed or sucked. The most of the water passes over the membrane, the undesired materials are held and they leave the system with the rest of the water, which flows tangentially without being filtered. The filtered water is called “permeate”, while the not filtered one, which has a high concentration of pollutants, is called “concentrate” or “reject”.
Membrane filtration processes can be used, in combination with each others or with other kinds of treatments, to remove ions, bacteria, microorganisms, particles, colloids and organic matter. Those pollutants give to the water undesired characteristics, making it not suitable for legislative purpose for the final user. Our aim is to use our experience and our know-how to identify which methods must be applied to solve in the best way the customer's problem, obtaining the best possible costs/benefits ratio.
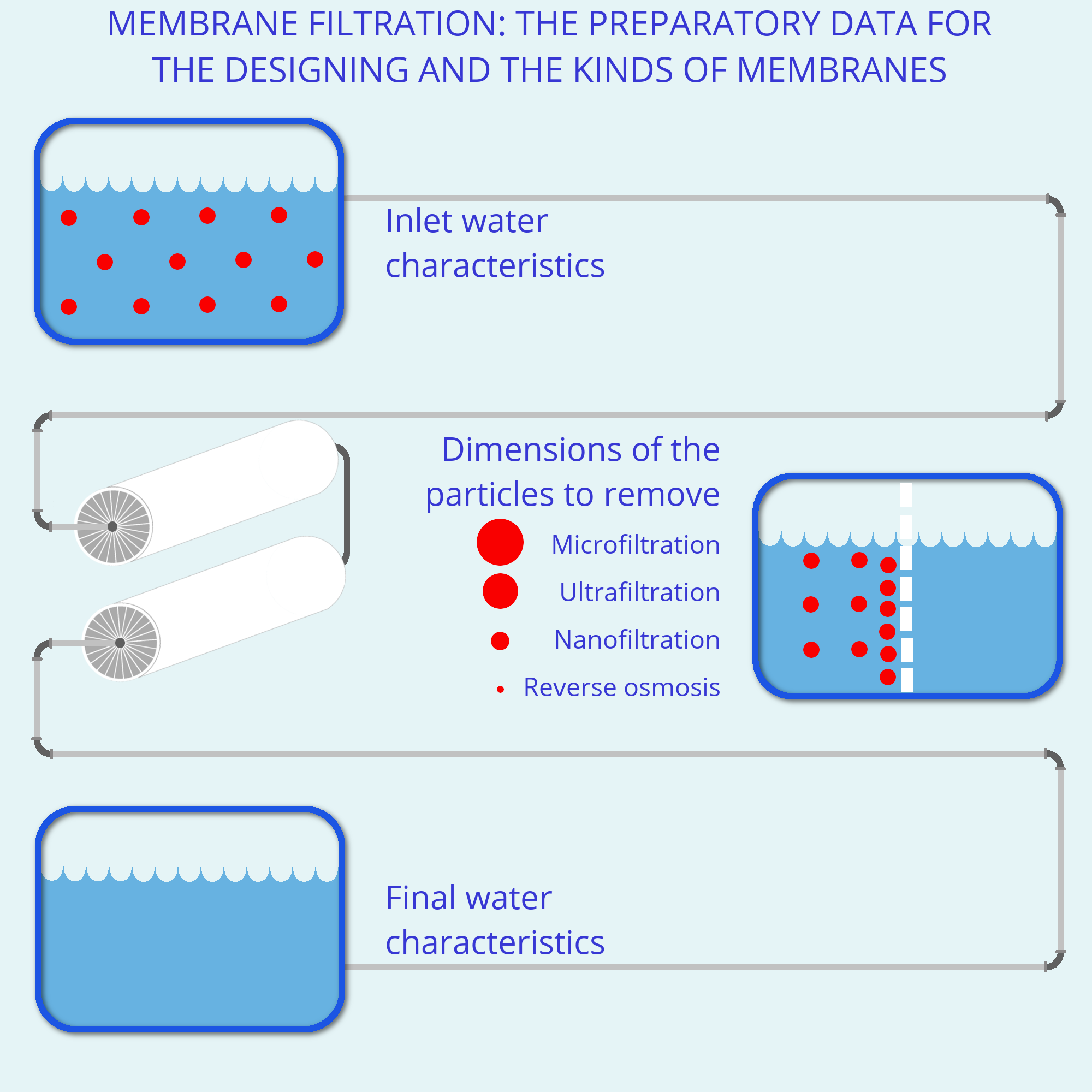
Membrane filtration's kinds
Depending on dimensions of the molecules which must be removed and, consequently, of the membrane pores, different kinds of membrane filtration can be distinguished: microfiltration (MF), nanofiltration (NF), ultrafiltration (UF) and reverse osmosis (RO).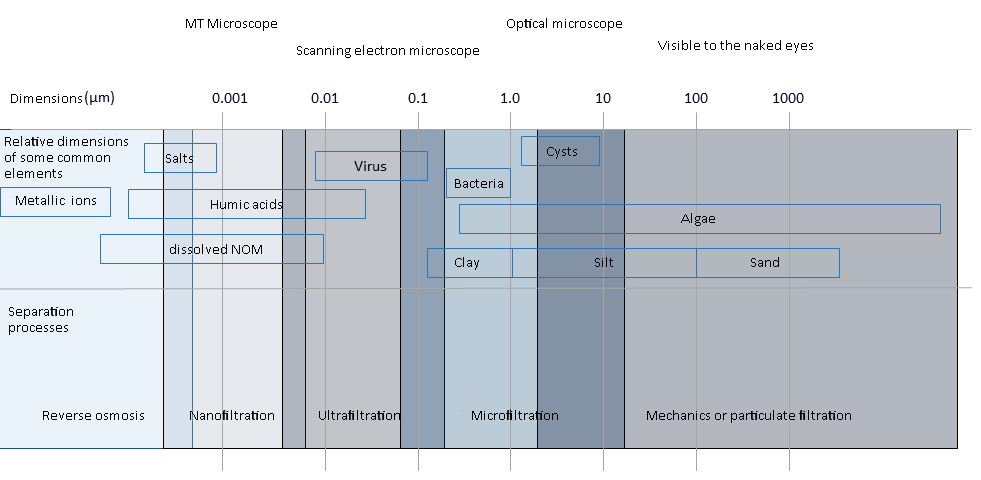
What is reverse osmosis (RO)?
Reverse osmosis, commonly abbreviated as RO, is the process in which the ions and the molecules are removed from a water solution, by applying pressure to the solution and making it to pass through a selective membrane. The natural osmosis process does not need energy and it consists in the passage of the water molecules through a selective membrane from a solution with a lower concentration to a solution with an higher concentration, following the so-called concentration gradient. The selective membrane allows the passage of water molecules, but not of ions (for example Na+, Ca2+, Cl-) or bigger molecules (for example glucose, urea and bacteria). Osmosis tends to continue until the two solutions have the same concentration of solutes (isotonic).
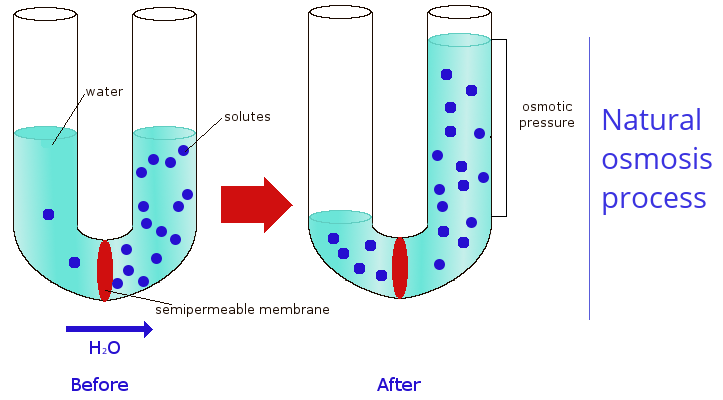
Reverse osmosis occurs when a pressure is exerted on the solution with an higher concentration to make the water to pass through the selective membrane to the solution with a lower concentration, so the movement goes against the concentration gradient. The pressure exerted must be higher than the natural osmotic pressure of the solution, which depends on its concentration.

Reverse osmosis is able to remove from the inlet water up to 99% of dissolved salts (ions), particles, colloids, organics, bacteria and pyrogens from the feed water, although a RO system should not be relied upon to remove 100% of bacteria and viruses. A RO membrane rejects contaminants on the basis of on their molecular size and ionic charge. Any contaminant that has a molecular weight greater than 200 or with a great ionic charge is likely rejected. Common membrane materials include polyamide thin film composites (TFC), cellulose acetate (CA) and cellulose triacetate (CTA). The membranes can be composed by material being spiral wound around a tube, or hollow fibres bundled together.
Reverse osmosis' applications:
- spring water treatments for civil and industrial uses;
- sea water treatments;
- wastewater treatments.
Reverse osmosis plants' architectures
Reverse osmosis plants can be designed as one or more stages plants and as one or more passes plants.
In a one stage RO system, the inlet water enters the system and a concentrate flow and a permeate flow exit from it. In a two-stage system the concentrate from the first stage becomes the feed water to the second stage. The permeate water collected from the first stage is combined with permeate water from the second stage. Additional stages increase the recovery from the system.
A RO plant's pass is a stand-alone RO system, so in a double pass RO plant, the permeate from the first pass becomes the inlet water for the second pass. In this way the final permeate will have a much higher quality.
Possible problems of reverse osmosis plants
The most common problems which a RO system may experience are fouling, scaling and chemical damages. Fouling occurs when contaminants accumulate on the membrane surface effectively plugging the membrane. This problem generates a higher pressure drop across the RO system and a lower permeate flow. Fouling can be caused by the presence of particulate, colloidal or organic matter, microorganism (bacteria, etc), which may product biofilms. Scaling occurs when certain inorganic dissolved compounds become more concentrated and they exceed their solubility limits and precipitate on the membrane surface as scale. The results of scaling are a higher pressure drop across the system, less salt rejection, low permeate flow and lower permeate water quality. The most common scale that forms on RO membranes is calcium carbonate (CaCO3).
Chemical damages are caused by the presence of oxidizers, such as chlorine or chloramines, which may generate holes in the membrane pores, creating irreparable damage. The result of chemical attack on an RO membrane is a higher permeate flow and a poorer quality permeate water. All of those problems can be solved with pre-treatment systems.
What is nanofiltration (NF)?
Nanofiltration membranes have pores with dimensions from 1 up to 5 nm and they can retain solid suspended matter with big dimensions, multivalent ions and solid suspended organic matter with low molecular weight. Those membranes have a effectively higher permeability than reverse osmosis membranes and so they can work with a lower pressure. Nanofiltration membrane are commonly thin film membranes (TFC), organised as spiral, tubular, or hollow modules (capillary), made of polyamide or cellulose acetate.
Nanofiltration membranes' properties are between the properties of reverse osmosis non porous membranes, in which the transport is caused by a diffusion mechanism, and the properties of ultrafiltration porous membranes, where the separation is caused by particles' dimensions and in some cases by their charge. The commercial nanofiltration membranes have a their own charge, due to the dissociation of some chemical groups on the surface, as carboxylic acids. Divalent ions can be removed by nanofiltration. Those ions are responsible of water hardness, for this reason nanofiltration is used for waters softening.
Problems of the nanofiltration membranes
Nanofiltration membranes are less vulnerable to fouling or scaling and pre-treatment is not necessary. Nanofiltration is often used as a pre-treatment method in reverse osmosis plants. Nanofiltration membranes cause the decrease of pH and preventive measures must be implemented, to avoid the water to become corrosive. Those measures can be mixing the produced water with the inlet water or adding some components to increase pH.
Nanofiltration's applications
The most common applications of nanofiltration are:
- water softening;
- organic substances, as pesticides and industrial colours, removing;
- total dissolved solids and total organic carbon decreasing;
- nitrates removing;
- heavy metals removing;
- pre-treatment.
What is ultrafiltration (UF)?
In the ultrafiltration separation mechanism membranes with pores with dimensions from 0.1 up to 0.001 micron are used. So the ultrafiltration is not able to remove organic molecules with low molecular weight and ions, like calcium, sodium, magnesium, sulphate and chloride. For this reason the differential osmotic pressure through the membrane surface is negligible. Low pressure values are sufficient to ensure high fluxes through the membrane. Ultrafiltration membranes modules can be plate – and – frame, wrapped in spirals or in tubular configurations. All those configurations are used for different applications.
Ultrafiltration's applications
The main applications of ultrafiltration are:
- removing of substances with high molecular weight, colloidal substances, organic and inorganic polymers, thin silt, bacteria and virus, including chlorine resistant like Cryptosporidium e Giardia;
- in place of classic secondary systems (coagulation, flocculation and sedimentation) and tertiary systems (sand filtration and chlorination);
- pre-treatment for reverse osmosis plants;
- final filtration for deionization plants.
What is microfiltration and its applications (MF)
Microfiltration membranes have the biggest pores among all the polymeric membranes, they have dimensions from 0.1 up to 10 micron. Microfiltration is used to separate the bigger suspended solids, as colloids, particles, fat and bacteria, while sugars, proteins, salts and all the molecules with low molecular weight pass through the membrane. Microfiltration membranes' pores have an asymmetric structure: with a tighter dimension near the surface to optimise the rejection and a wider one in the membrane section to optimise the flux.
Microfiltration is mainly used as pre-treatment in RO or NF plants to remove synthetic or natural organic material, reducing the encrust capability.
Our plants
Installation place: Mozambico
Plant flow rate: 20 mc/h
Problem to solve: necessity to have drinking water in zones where it's not possible to install an unmoveable plant in the settlements.
Kind of plant: potabilization plant in an insulate shelter of 40' composed by: multimedia filtration plant with pyrolusite, quartzite and activated carbons, ultrafiltration plant with n. 3 membranes for a total membrane surface of 150 mq, reverse osmosis plant with n. 8 membranes, UV lamps and relative dosing systems. The plant is equipped with automatic measurement devices for all the necessary parameters (pH indicators, flux indicators, etc...). A flux and maintenance station have been inserted. Plant's work is controlled by a PLC specially programmed by us.
Installation place: Castiglion della Pescaia (GR)
Plant flow rate: 114 mc/day
Problemto solve: the necessity to have fresh water in an accommodation facility by reliable plants, with low energy consumption and small acoustic impact.
Kind of plant: n. 2 reverse osmosis desalination plants in insulate container, including pre filtration plant of the inlet water, automatic dosing for the anti precipitant and all the other components for the reduction of the energy consumption, power electric, control and management plants. All the main parameters, which determine the plant correct functioning, are measured in an automatic way. The relative measurement devices are connected with the PLCs, which allow their control and management, both in the field and in remote.
Our customers
PPM offers its services to:
- mechanic industry;
- chemical industry;
- paper industry;
- ceramic industry;
- pharmaceutical and cosmetic industry;
- galvanic industry;
- wood industry;
- leather and textile industry;
- petrochemical industry;
- plastic industry;
- agro – food industry;
- painting and graphic industry;
- car wash;
- Petrol stations;
- Urban waste water treatment for integrated water services, public authorities and large or tourist establishment.
How do we work?

The first work phase is the collection of all the necessary data for designing of the best and more efficient solution for the customer. Those data include the inlet water quality, its final characteristics and the evaluation of customer's other needs. Chemical and physical analysis must be done to evaluate inlet water characteristics and sometimes it's also necessary a visit.
In the following phase data are evaluated and studied to proceed with the water treatment plant designing. In the case of the membrane filtration which kind of membrane is the most suitable to remove the present pollutants is determined. On the basis of the flux and on the desired permeate quality, the plant architecture is determined, i.e. how many membranes are to be installed and in which passes or stages. Then the plant is constructed, assembling the different components of the best quality. The plant is installed and tested in its final position.







